F. Christopher Pigge
Keywords
- Organometallics in organic synthesis
- Synthesis of heterocyclic compounds related to alkaloids
- Design and synthesis of heterocyclic cyclophanes
- Supramolecular host-guest chemistry
- Crystal engineering
Focus areas
- Natural products/medicinal
- Synthesis
Research interests
We seek to utilize our expertise in synthetic chemistry to 1) develop new strategies for organic synthesis; 2) design and construct new bio-conjugation reagents for applications in radiochemistry and radiopharmaceuticals; and 3) explore supramolecular approaches to functional materials.
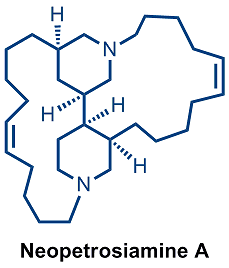
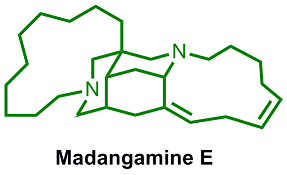
We have a longstanding interest in utilizing dearomatization as a tactic for accessing stereochemically rich small-molecule synthetic building blocks from easily prepared aromatic precursors. Organometallic activation as a means to achieve concomitant dearomatization and C-C bond formation offers concise and stereocontrolled approaches to diverse bi- and polycyclic ring systems. Construction of architecturally sophisticated heterocyclic ring systems via manipulation of heteroarene precursors is also under active investigation. We seek to uncover concise and stereocontrolled routes to a variety of medicinally relevant azacyclic derivatives and natural product frameworks, such as those encountered in bis(piperidine) alkaloids (e.g., neopetrosiamine A) and the madangamine alkaloids.
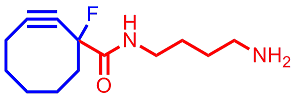
Radiopharmaceuticals offer unparalleled sensitivity for in vivo diagnostic imaging of cancer and other diseases. With the aim of developing general methods for the synthesis of radionuclide chelators conjugated to bio-targeting molecules, we have designed several bifunctional bio-conjugation agents that feature strained cycloalkyne moieties. Assembly of bio-conjugated radiochelators can then be achieved via application of metal-free click chemistry with azide-modified azamacrocycles or azide-modified targeting ligands (such as peptides or oligoRNA sequences).
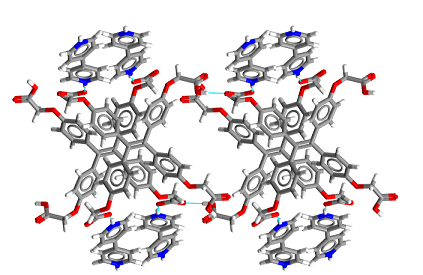
The synthesis of functional materials through non-covalent bonding is also emphasized in the Pigge group. Our approach entails identification of organic building blocks that possess inherent properties on the molecular level that can be expressed at the supramolecular level. Specifically, organic compounds that exhibit luminescence and/or redox activity in aggregated states are being investigated for incorporation into larger (ideally crystalline) supramolecular assemblies (both purely organic as well as metal-organic frameworks). Materials prepared from these organic components will then be imbued with desirable bulk properties (such as porosity, fluorescence, and conductance) relevant to potential applications in molecular electronics, chemical sensing, and catalysis. For example, shown below is a crystalline organic semiconducting material assembled from a tetraarylethylene derivative and 4,4’bipyridine.
Recent publications
- "A Turn-On AIE Active Fluorescent Sensor for Hg2+ by combination of 1,1-Bis(2-pyridyl)ethylene and Thiophene/Bithiophene Fragments.” Moustafa T. Gabr and F. Christopher Pigge, Mater. Chem. Front. 2017, 1, 1654-1661.
- Synthesis and Evaluation of Tetraarylethylene-based Mono-, Bis-, and Tris(pyridinium) Derivatives for Image-Guided Mitochondria-Specific Targeting and Cytotoxicity of Metastatic Melanoma Cells.” Jessica L. Reedy, Devin K. Hedlund, Moustafa T. Gabr, Grant M. Henning, F. Christopher Pigge, and Michael K. Schultz, Bioconjugate Chem. 2016, 27, 2424-2430.
- "Construction of 1,2,4-Triazole Derivatives via Cyclocondensation of Alkylidene Dihydropyridines and Aryldiazonium Salts." Madhur S. Joshi and F. Christopher Pigge, Org. Lett. 2016, 18, 5916-5919.
- “Strain-Promoted Cycloadditions for Development of Copper-Free Click Reactions.” F. Christopher Pigge, Curr. Org. Chem. 2016, 20, 1902-1922.
- ‘’A Selective Fluorescent Sensor for Zn2+ based on Aggregation-Induced Emission (AIE) Activity and Metal Chelating Ability of Bis(2-pyridyl)diphenylethylene.’’ Moustafa T. Gabr and F. Christopher Pigge, Dalton Trans. 2016, 45, 14039.
- ‘’Alkylidene Dihydropyridines as Synthetic Intermediates: Model Studies toward the Synthesis of the Bis(piperidine) Alkaloid Xestoproxamine C.’’ Ashabha I. Lansakara, S. V. Santhana Mariappan and F. Christopher Pigge, J. Org. Chem. 2016, 81, 10266-10278.
- ‘’Dearomatized Alkylidene Dihydropyridines as Substrates for Mizoroki-Heck Cyclizations.’’ Madhur S. Joshi and F. Christopher Pigge, ACS Catalysis. 2016, 6, 4465-4469.
- ‘’Synthesis and Aggregated-Induced Emission Properties of Pyridine and Pyridinium Analogues of Tetraphenylethylene.’’ Moustafa T. Gabr and F. Christopher Pigge, RSC Adv. 2015, 5, 90226-90234.
- ‘’Charge-Assisted Halogen Bonding in Bromo- and Iodophenylpyridinium Chlorides.’’ Christopher J. Kassl, Dale C. Swenson and F. Christopher Pigge, Cryst. Growth Des. 2015, 15, 4571-4580.
- "Intramolecular Cyclization of Alkylimidazoles." Madhur S. Joshi, Ashabha I. Lansakara, and F. Christopher Pigge, Tetrahedron Lett. 2015, 56, 3204-3207.
- “Anion Detection by Aggregation-Induced Enhanced Emission (AIEE) of Urea-Functionalized Tetraphenylethylenes.” Christopher J. Kassl and F. Christopher Pigge, Tetrahedron Lett. 2014, 55, 4810-4813.
- “Brønsted Acid Catalyzed Intramolecular Benzylic Cyclizations of Alkylpyridines.” Ashabha I. Lansakara, Daniel P. Farrell, and F. Christopher Pigge, Org. Biomol. Chem. 2014, 12, 1090-1099.
- “Gold-Catalyzed Intramolecular Cyclization of Alkynones with Pyridine Anhydrobases.” Lokesh Pawar and F. Christopher Pigge, Tetrahedron Lett.2013, 54, 6067-6070.
- “Harnessing Aggregation Induced Emission (AIE) of Tetraphenylethylenes in Metal Ion Sensing and Luminescent Metal-Organic Frameworks.” F. Christopher Pigge, J. Mol. Eng. Mater. 2013, 1, 1340009
- "Halogen Bonded Networks from Pyridyl-Substituted Tetraarylethylenes and Diiodotetrafluorobenzenes.” F. Christopher Pigge, Pradeep P. Kapadia, and Dale C. Swenson, CrystEngComm 2013, 15, 4386-4391.
- “MRI Markers for MRI-Guided HDR Brachytherapy: Novel Marker-Flange for Cervical Cancer and Marker-Catheters for Prostate Cancer.” Joshua Schindel, Manickam Muruganandham, F. Christopher Pigge, James Anderson, and Yusung Kim, Int. J. Radiation Oncol. Biol. Phys. 2013, 86, 387-393.
- “Intramolecular Cyclization Manifolds of 4-Alkylpyridines Bearing Ambiphilic Side Chains: Construction of Spirodihydropyridines or Benzylic Cyclization via Anhydrobase Intermediates.” Sharavathi G. Parameswarappa and F. Christopher Pigge, J. Org. Chem. 2012, 77, 8038-8048
- “Influence of Halogen Bonding Interactions in Crystalline Networks of Tetraarylethylene Halobenzoyl Esters.” Pradeep P. Kapadia, Dale C. Swenson, and F. Christopher Pigge, Cryst. Growth Des. 2012, 12, 698-706.
- “Improved Synthesis and Biological Evaluation of Chelator-Modified a-MSH Analogues Prepared by Copper-Free Click Chemistry.” Nicholas J. Baumhover, Molly E. Martin, Sharavathi G. Parameswarappa, Kyle C. Kloepping, M. Sue O’Dorisio, F. Christopher Pigge, and Michael K. Schultz, Bioorg. Med. Chem. Lett. 2011, 21, 5757-5761.
- “Generation and Solid State Characterization of Tetrapyridinium Perchlorate Salts Derived from Tetrapyridyl Tetraphenylethylenes.” Pradeep P. Kapadia, Mackenzie A. Magnus, Dale C. Swenson, and F. Christopher Pigge, J. Mol. Struct. 2011, 1003, 82-86.
- “Synthesis of Substituted 3,9-Diazaspiro[5.5]undecanes via Spirocyclization of Pyridine Substrates.” Sharavathi G. Parameswarappa and F. Christopher Pigge, Tetrahedron Lett. 2011, 52, 4357-4359.
- “Semiconducting Organic Assemblies Prepared from Tetraphenylethylene Tetracarboxylic Acid and Bis(pyridine)s via Charge-Assisted Hydrogen Bonding.” Pradeep P. Kapadia, Lindsay R. Ditzler, Jonas Baltrusaitis, Dale C. Swenson, Alexei V. Tivanski, and F. Christopher Pigge, J. Am. Chem. Soc. 2011, 133, 8490-8493.
- “Tetrapyridyl Tetraphenylethylenes: Supramolecular Building Blocks with Aggregation Induced Emission Properties.” Pradeep P. Kapadia, John C. Widen, Mackenzie A. Magnus, Dale C. Swenson, and F. Christopher Pigge, Tetrahedron Lett. 2011, 52, 2519-2522.
- “Losing Control? “Design” of Crystalline Organic and Metal-Organic Networks using Conformationally Flexible Building Blocks.” F. Christopher Pigge, CrystEngComm 2011, 13, 1733-1748. [Invited review]
- “Titanium-Mediated Spirocyclization Reactions of 4-Alkyl Pyridines.” Sharavathi G. Parameswarappa and F. Christopher Pigge, Org. Lett. 2010, 12, 3434-3437.
- “A DOTA-Peptide Conjugate by Copper-Free Click Chemistry.” Molly E. Martin, Sharavathi G. Parameswarappa, M. Sue O’Dorisio, F. Christopher Pigge, and Michael K. Schultz, Bioorg. Med. Chem. Lett. 2010, 20, 4805-4807.
- “Synthesis of a DOTA-Biotin Conjugate for Radionuclide Chelation via Cu-Free Click Chemistry.” Michael K. Schultz, Sharavathi G. Parameswarappa, and F. Christopher Pigge, Org. Lett. 2010, 12, 2398-2401.
- “Metal Catalyzed Allylation of Organoboranes and Organoboronic Acids.” F. Christopher Pigge, Synthesis 2010, 1745-1762. [Invited review]
- “Examination of Halogen Bonding Interactions in Electronically Distinct but Structurally Related Tris(haloarenes).” F. Christopher Pigge, Venu R. Vangala, Dale C. Swenson, and Nigam P. Rath, Cryst. Growth Des. 2010, 10, 224-231.
- “Synthesis of Polycyclic Cyclohexadienyl Ruthenium(II) Complexes from h6-Arene Precursors via Phosphine Promoted Intramolecular Nucleophilic Aromatic Addition.” F. Christopher Pigge, R. Dhanya, and Dale C. Swenson, Organometallics 2009, 28, 3869-3875.
- “A 1D Crystalline Organic Polyrotaxane Formed via Hydrogen Bond-Mediated Self-Assembly of a Conformationally Flexible Tri-Carboxylic Acid.” F. Christopher Pigge, Mayuri K. Dighe, and Dale C. Swenson, CrystEngComm 2009, 11, 1227-1230.
- Organic chemistry
