David F. Wiemer
Postdoctoral research fellowships
- Alfred P. Sloan Foundation Fellow (1985-89)
- NIH Postdoctoral Fellowship, Cornell University (1976-78)
Keywords
- Organophosphorus
- Phosphoantigen
- Enzyme inhibitors
Research interests
Our research interests include development of new strategies for organic synthesis based on organophosphorus compounds, design and synthesis of metabolic probes, and total synthesis of biologically active natural products.
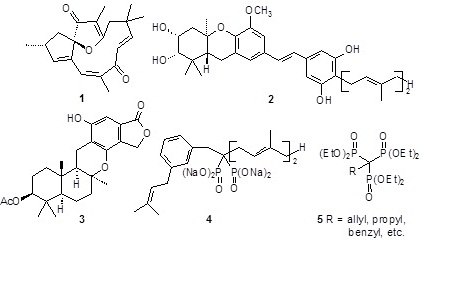
The natural products we target often are terpenoids with anticancer potential. Past goals included (+)-jatrophone (1), while more recent targets include agents such as schweinfurthin A (2) and kampanol (3) which display interesting profiles of biological activity. Through collaboration with the National Cancer Institute, the activity of our products is determined and new analogs are designed.
The synthesis of complicated targets often requires development of new approaches to the formation of a particular substructure. This is part of the challenge, and the opportunity, of synthetic chemistry. A major focus of our efforts has been development of new methods for formation of carbon-phosphorus bonds and new applications of organophosphorus compounds in organic synthesis. For example, we have explored use of electrophilic phosphorus reagents for preparation of otherwise inaccessible phosphonates. These methodologies lead to new strategies for facile assembly of complex molecules and become key parts of our total syntheses. They also can be applied in preparation of novel enzyme inhibitors.
Our interests in terpenoid synthesis and organophosphorus chemistry have combined in several investigations of isoprenoid metabolism. In collaboration with Prof. Raymond J. Hohl (Internal Medicine and Pharmacology) we have designed and prepared novel terpenoid bisphosphonates (4) that serve as inhibitors of geranylgeranyl diphosphate synthase. In collaboration with Prof. Sarah A. Holstein (Internal Medicine) we are working to develop potent and selective inhibitors of the downstream enzyme geranylgeranyl transferase II. Through synthesis and bioassay of isoprenoid pyrophosphate analogues, we hope to clarify their value as potential anti-cancer agents and learn how to modulate isoprenoid metabolism. These studies also draw on our interest in C-P bond formation to generate new motifs for phosphorus-containing enzyme inhibitors. For example, we recently reported synthesis of alkyl-1,1,1-trisphosphonates (5), and have begun to explore the chemistry of this system.
Recent publications
- Zhou, X.; Hartman, S.; Born, E. J.; Smits, J. P.; Holstein, S. A.; Wiemer, D. F., Triazole-based inhibitors of geranylgeranyl transferase II. Bioorg. Med. Chem. Lett. 2013, 23, 764–766.
- Richardson, R. M.; Wiemer, D. F., Direct Conversion of Benzylic and Allylic Alcohols to Phosphonates, Organic Syntheses 2013, 90, 145–152.
- Richardson, R. M.; Barney, R. J.; Wiemer, D. F., Synthesis of dialkyl and diaryl benzylphosphonates through a ZnI2-mediated reaction. Tetrahedron Lett. 2012, 53, 6682–6684.
- Kuder, C. H.; Neighbors, J. D.; Wiemer, D. F.; Hohl, R. J., Functional evaluation of a fluorescent schweinfurthin: Mechanism of cytotoxicity and intracellular quantification. Mol. Pharm. 2012, 82, 9-16.
- Topczewski, J. J.; Callahan, M. P.; Kodet, J. G.; Inbarasu, J. D.; Mente, N. R.; Beutler, J. A.; and Wiemer, D. F., Relevance of the C-5 Position to Schweinfurthin Induced Cytotoxicity. Bioorg. Med. Chem. 2011, 19, 7570–7581.
- Smits, J.P.; Wiemer, D.F., Synthesis and Reactivity of Alkyl-1,1,1-trisphosphonate Esters. J. Org. Chem. 2011, 76, 8807–8813.
- Wiemer, A.J.; Wiemer, D.F.; Hohl, R.J., Geranylgeranyl diphosphate synthase: an emerging therapeutic target. Nature: Clinical Pharmacology and Therapeutics, 2011, 90 805–812
- Barney, R. J.; Richardson, R. M.; Wiemer, D. F., Direct conversion of benzylic and allylic alcohols to phosphonates. J. Org. Chem. 2011, 76, 2875–2879.
- A. Topczewski, J. J.; Kodet, J. G.; Wiemer, D. F., Exploration of Cascade Cyclizations Terminated By Tandem Aromatic Substitution: Total Synthesis of (+)-Schweinfurthin. J. Org. Chem. 2011, 76, 909–919.
- Ulrich, N. C.; Kuder, C. H.; Hohl, R. J.; Wiemer, D. F., Biologically active biotin derivatives of schweinfurthin F. Bioorg. Med. Chem. Letters, 2010, 20, 6716–6720.
- Topczewski, J. J.; Callahan, M. P.; Neighbors, J. D.; Wiemer, D. F. A Tandem Cascade Cyclization-Electrophilic Aromatic Substitution: Application in the Total Synthesis of (+)-Angelichalcone. J. Am. Chem. Soc. 2009, 131,14630–14631.
- Topczewski, J. J.; Neighbors, J. D.; Wiemer, D. F. Total Synthesis of (+)-Schweinfurthins B and E. J. Org. Chem. 2009, 74, 6965–6972.
- Wiemer, A. J.; Yu, J. S.; Shull, L. W.; Barney, R. J.; Wasko, B. M.; Lamb, K. M; Hohl, R. J.; Wiemer, D. F. Pivaloyloxymethyl-modified isoprenoid bisphosphonates display enhanced inhibition of cellular geranylgeranylation. Bioorg. Med. Chem. 2008, 16, 3652–3660.
- Wiemer, A. J.; Yu, J. S.; Lamb, K. M; Hohl, R. J.; Wiemer, D. F. Mono- and Dialkyl Isoprenoid Bisphosphonates as Geranylgeranyl Diphosphate Synthase Inhibitors. Bioorg. Med. Chem. 2008, 16, 390–399.
- Mente, N. R.; Neighbors, J. D.; Wiemer, D. F. BF3·Et2O Mediated Cascade Cyclizations: Synthesis of Schweinfurthins F and G. J. Org. Chem. 2008, 73, 7963–7970.
- Organic chemistry
